Photo courtesy of Respiree
Singapore's Health Sciences Authority has approved an AI-powered software that identifies patient deterioration.
Singaporean startup Respiree's 1Bio AI-Acute toolbox has been cleared as a Class B software as a medical device, the first approval of its kind in the country for an AI-powered alternative to an inpatient early warning score.
HOW IT WORKS
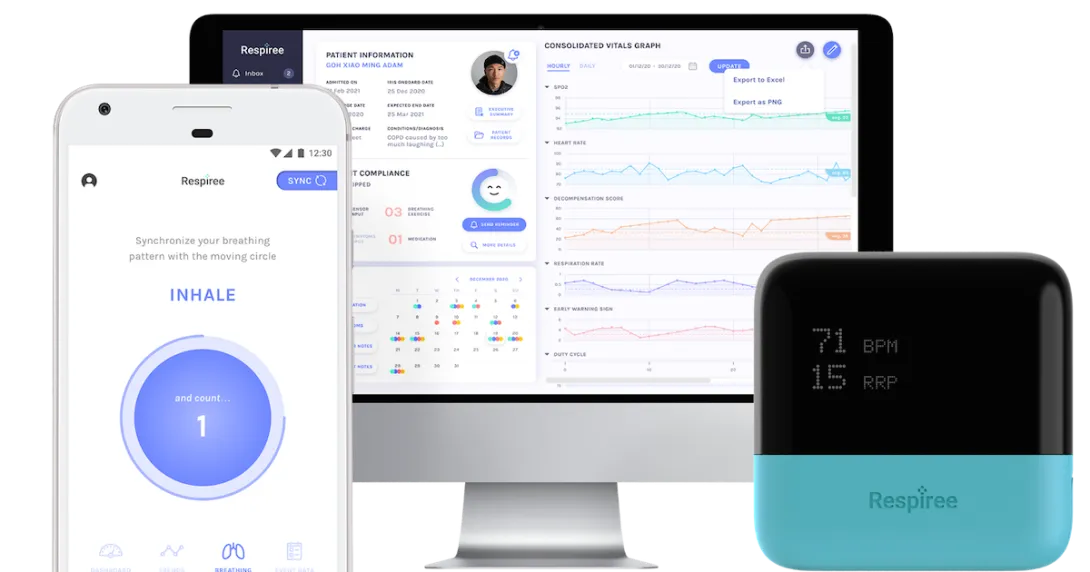
The AI software generates a probability score of an impending acute deterioration, defined as an "unplanned ICU transfer" or a "rapid response." A score is displayed as a visual trend on the 1Bio Platform dashboard.
"To explain the change in score, physicians can plot the corresponding vital signs and visualise the correlation in change in vitals trend with the 1Bio AI score itself," Respiree CEO and founder Dr Gurpreet Singh explained to Mobihealth News.
The platform-agnostic AI can ingest data, including bedside vitals and EMR, from any devices.
The AI toolbox is integrated into the Respiree 1Bio platform, which is also HSA-approved, along with the RS001 wearable cardio-respiratory sensor device. The HSA approval also followed a validation study with Mayo Clinic.
Respiree mentioned in its statement that it is now preparing to file for similar approvals across Asia, Australia, New Zealand, and the United States "in the coming months." "These are ongoing efforts," Dr Singh said. The company previously announced plans to also enter markets across the Gulf Cooperation Council states and Thailand.
"Deployments and pilots are underway at the moment," Dr Singh added, without specifying where. Respiree is reportedly collaborating with Roche Diagnostics on pilot projects across the Asia-Pacific.
WHY IT MATTERS
Respiree claims its solution to be more precise in detecting acute deteriorations than current models. Dr Singh notes how current threshold-based methods often have low precision, resulting in more unnecessary alarms and notifications.
The company plans to develop AI toolboxes for more disease-specific conditions, like sepsis, according to Dr Singh. "For now, the intended use [of the 1Bio AI] is for general deterioration prediction."
THE LARGER TREND
Early this year, Respiree obtained an additional approval from the Singapore HSA for the expanded use of its wearable monitoring device for children. In August, it received another 510(k) clearance from the United States Food and Drug Administration, also for an expanded use of its device at home, which covers the 1Bio Platform. Besides Singapore and the US, RS001 is also approved in Australia and across Europe. Moreover, it has been incorporated in hospital and district-wide remote patient monitoring projects in Singapore and Australia.
Respiree also recently closed a $11.6 million Series A funding round in July.